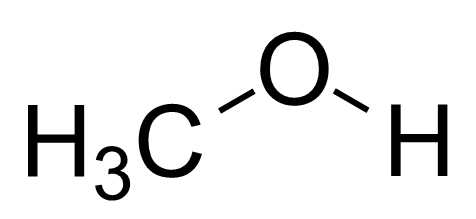
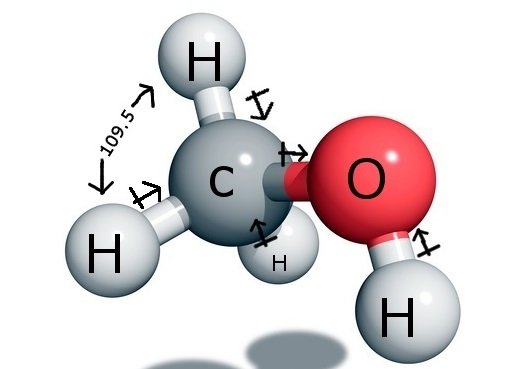
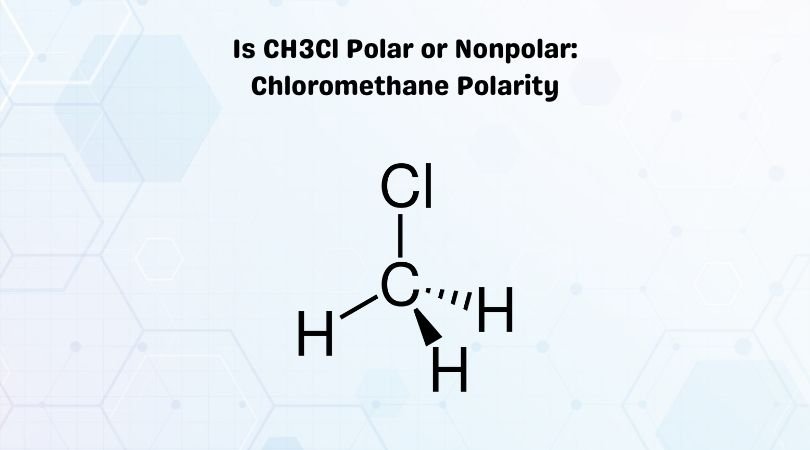
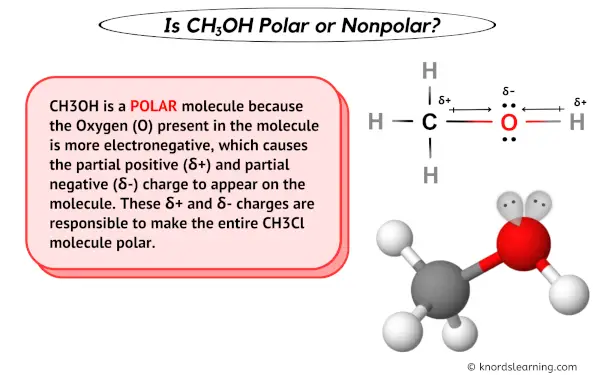
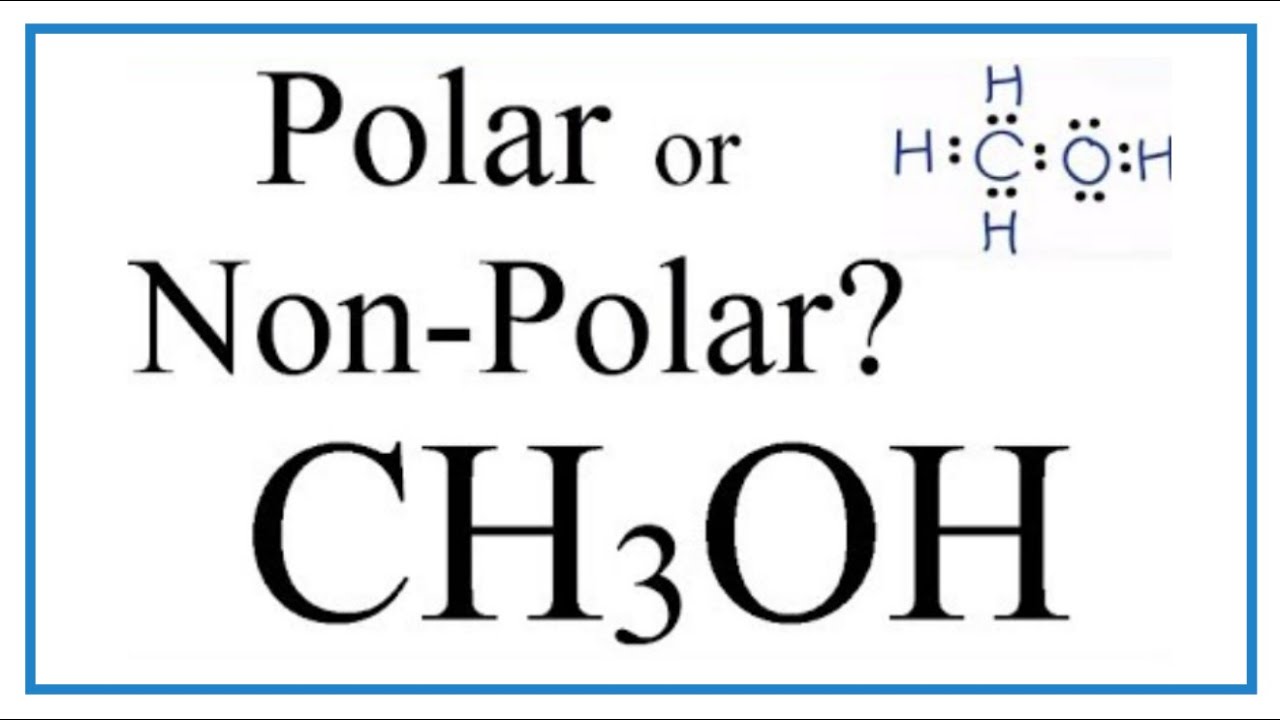
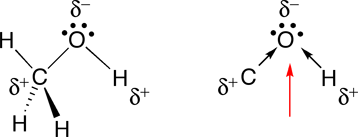Is CH3OH Polar or Nonpolar? - Polarity of Methanol | Polar, Covalent bonding, Electron configuration

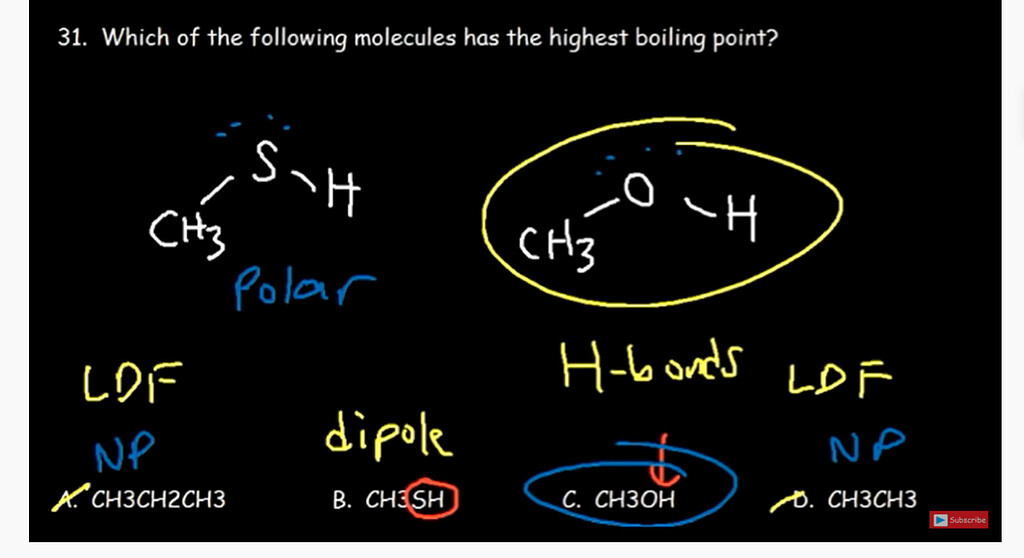
SOLVED: Which of the following has the highest boiling point? a) CH3OH ( polar) b) C2H6 (nonpolar) c) HCl d) O2 Which of the following has the highest boiling point? (They are all