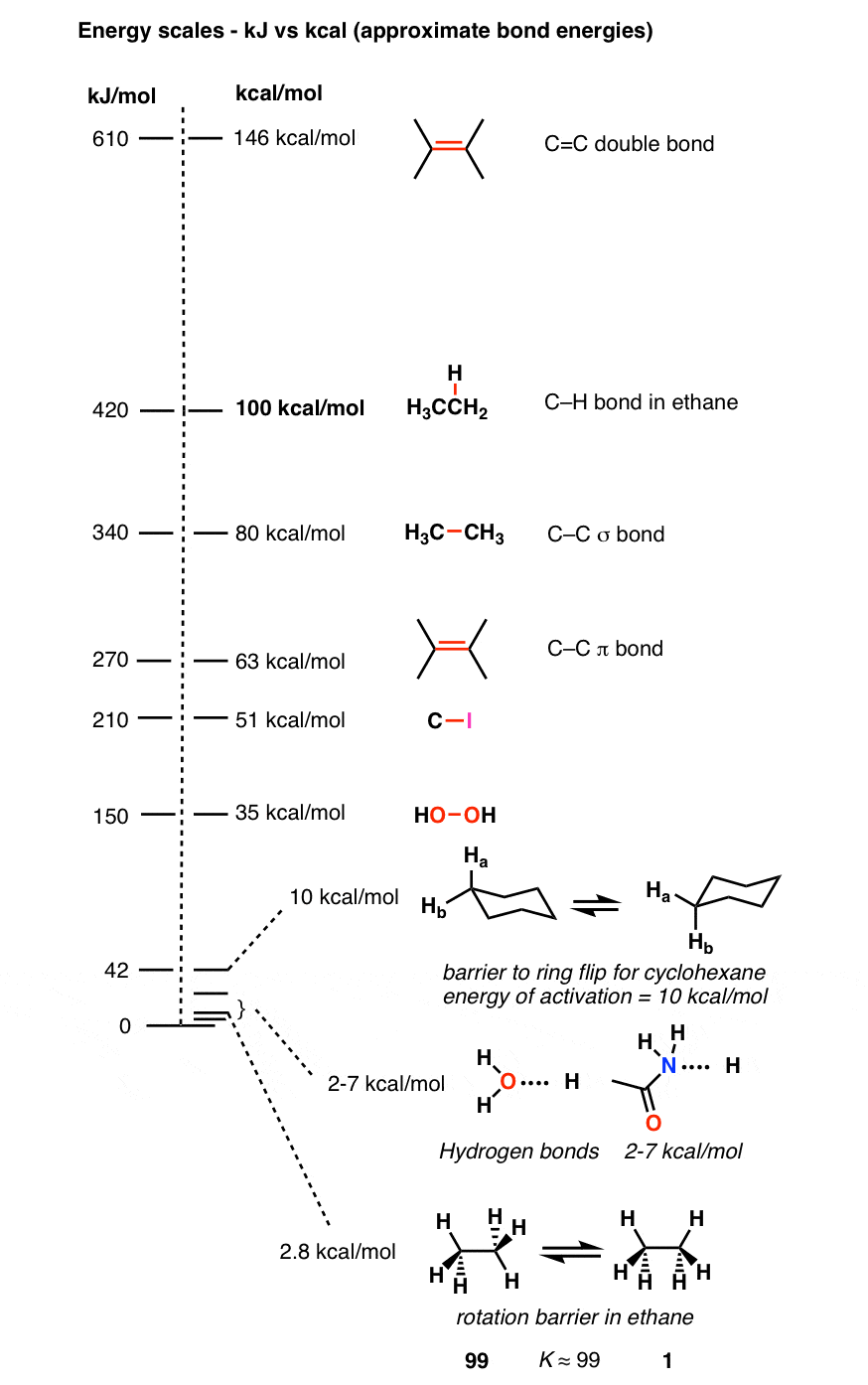
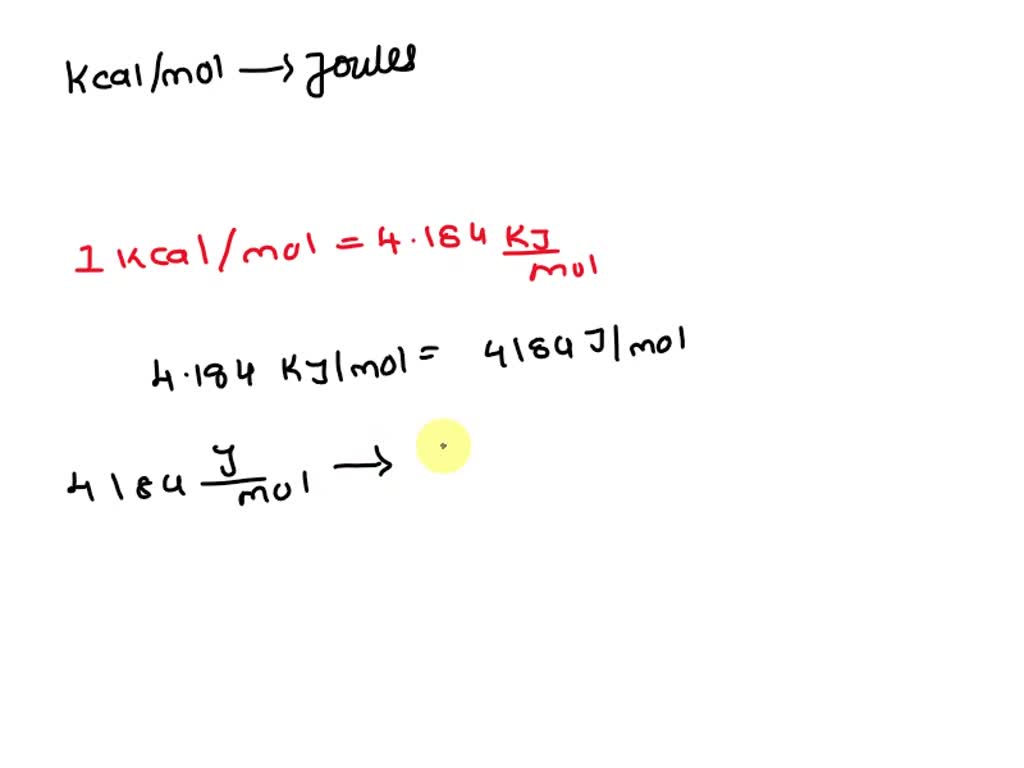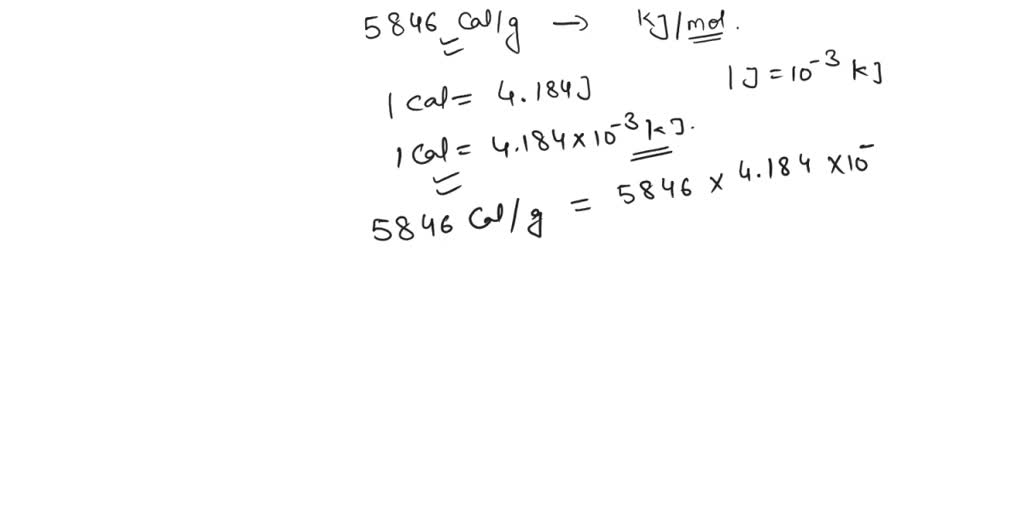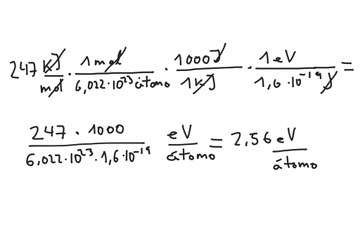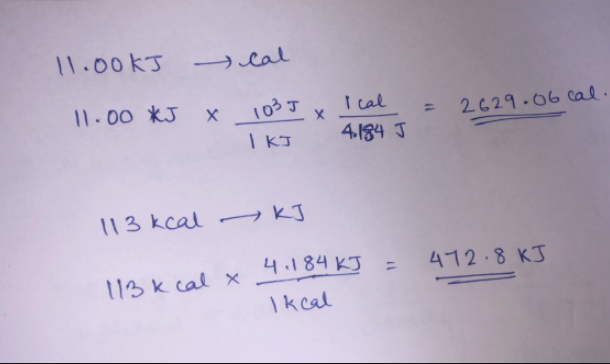SOLVED: The energy associated with chemical processes is often quoted in kilojoules per mole of reactants. Please derive the conversion factor from eV/molecule to kJ/mol. (1 eV = 60218 × 10-19 J, 1 mole = 6.02214076 × 1023).
Is Kd (the dissociation constant) for a given protein-ligand pair directly correlated to binding affinity (kcal/mol, Rosetta Energy Units, etc.)? - Quora

SOLVED: Convert these two atomic units (a.u.) into kJ/mol by using 2625 kJ/ mol = 1 a.u. -78.7939 kJ/mol -78.7838378 kJ/mol Please show the work.
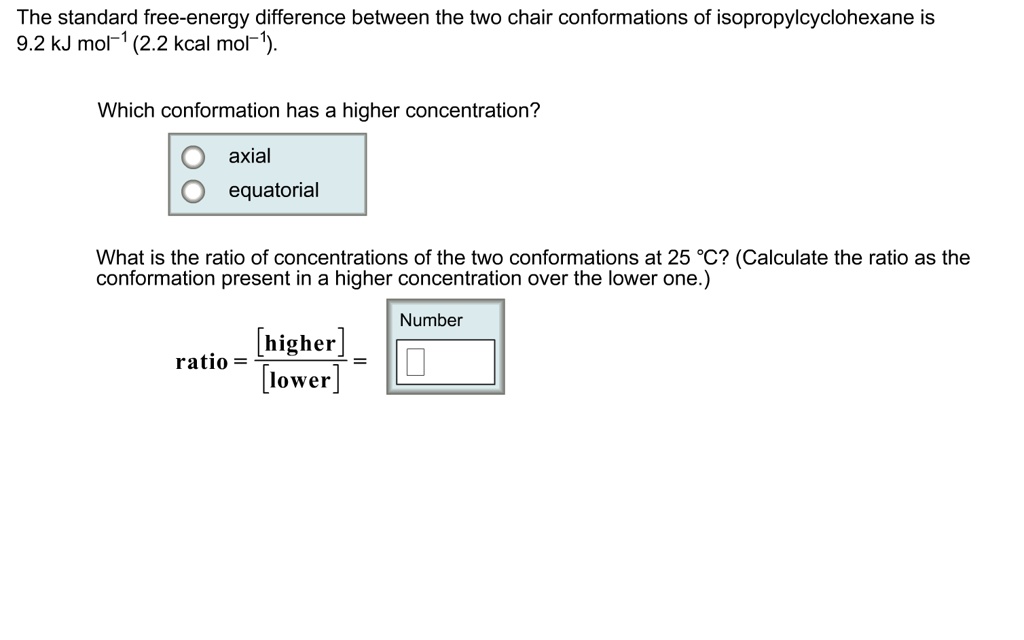
SOLVED: The standard free-energy difference between the two chair conformations of isopropylcyclohexane is 9.2 kJ mol- (2.2 kcal mol Which conformation has a higher concentration? axial equatorial What is the ratio of

The value of universal gas constant is R=8.3 J/kcal/mol. The value of R is atmosphere litre per kelvin/mol is?

Table 2 from A mathematical introduction to Hartree-Fock SCF methods in Quantum Chemistry | Semantic Scholar

Thermodymanics Lecture 3 8/31/2004. Units Energy Ultimate source of energy is the sun E = h 57 Kcal/mol of photons green light or 238 KJ/mol 1 cal = - ppt download