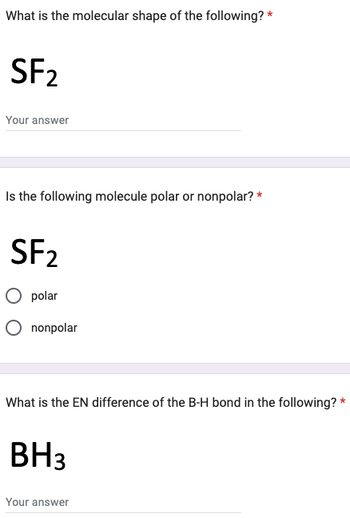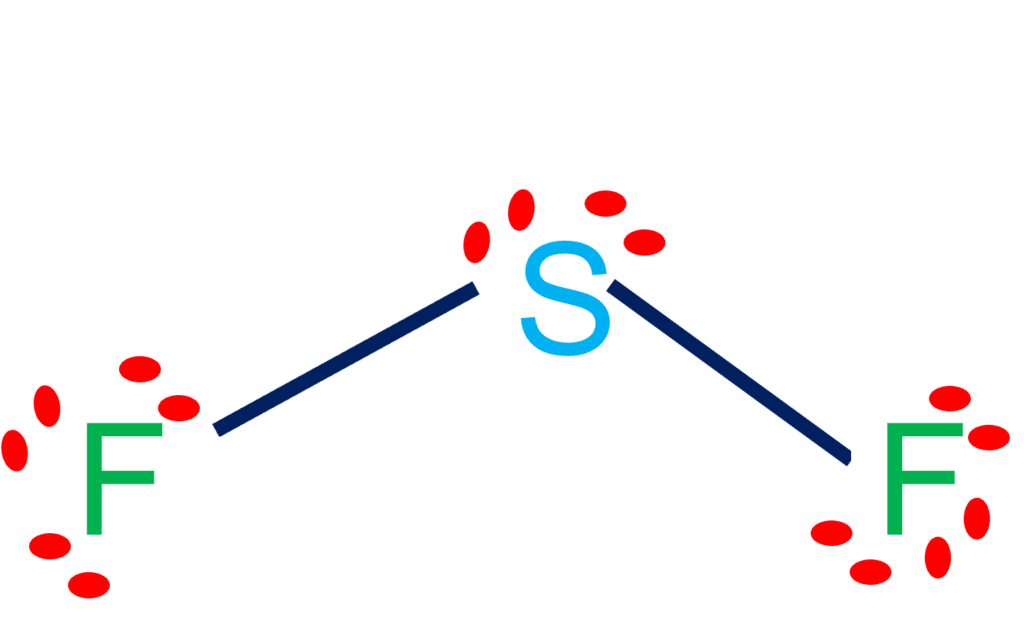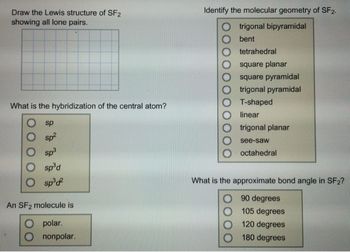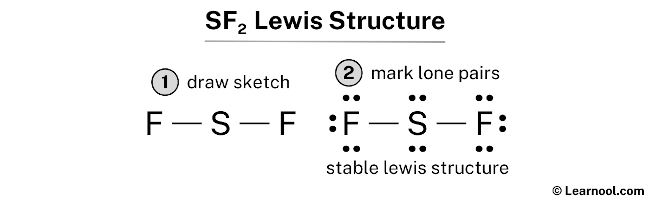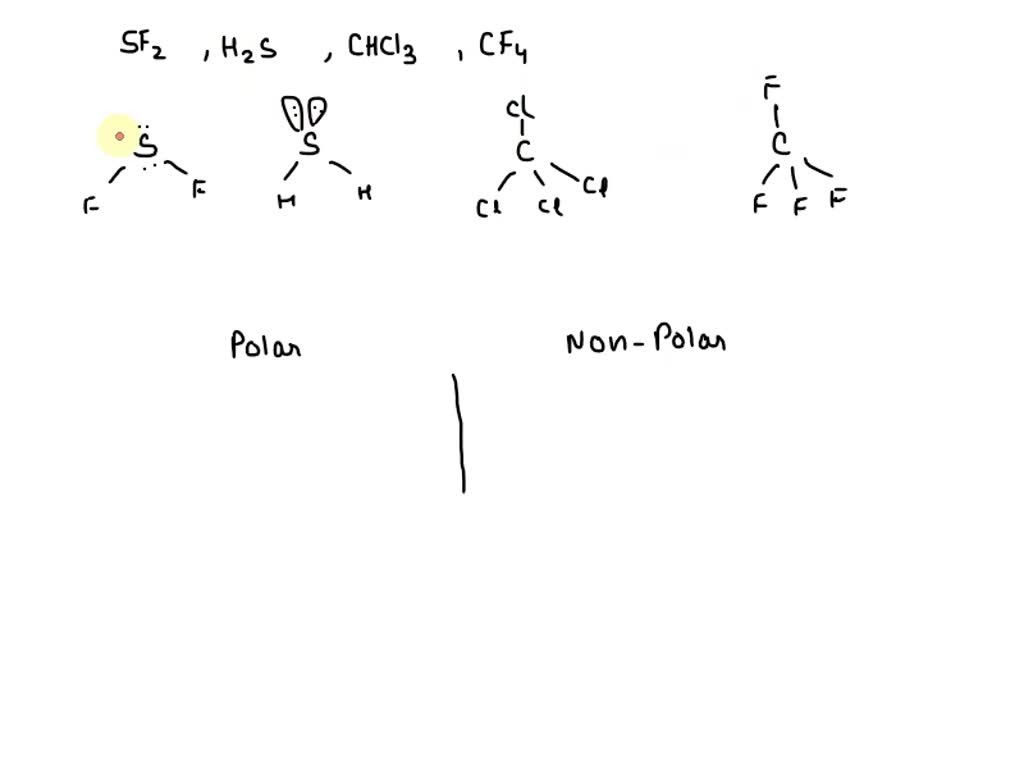
SOLVED: Classify the bonds of the following molecules as polar or nonpolar: Drag the appropriate items to their respective bins. sulfur difluoride (SF2) hydrogen sulfide (H2S) chloroform (CHCl3) carbon tetrafluoride (CF4) Polar

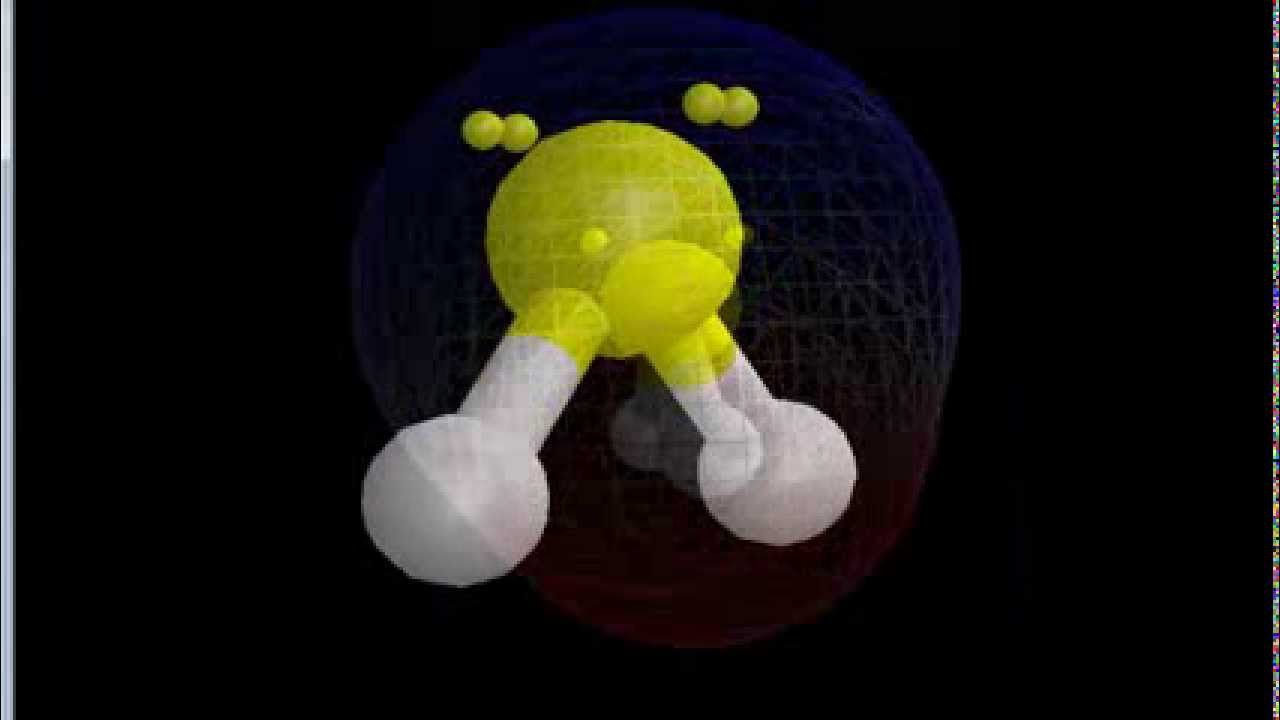
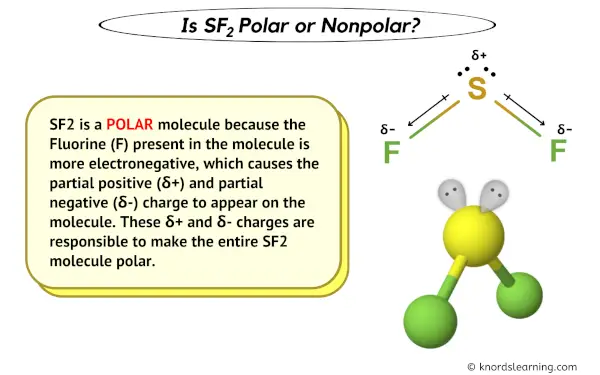
Is SF2 Polar or Nonpolar? - Polarity of Sulfur difluoride | Electron configuration, Molecular geometry, Polar